Korean developers take part in global boom in obesity treatments
Published: 13 Apr. 2026, 11:39
![Weight loss medications, Wegovy, front, and Saxenda, are placed on a hand at a pharmacy in central Seoul on Dec. 2, 2024. [YONHAP]](https://koreaseafood.online/data/photo/2026/04/13/51957417-906a-4ed6-b04e-04b9876c58df.jpg)
Weight loss medications, Wegovy, front, and Saxenda, are placed on a hand at a pharmacy in central Seoul on Dec. 2, 2024. [YONHAP]
The global boom in obesity treatments has evolved into a race centered on edible weight loss medications among pharmaceutical companies worldwide — and Korean developers are also taking part.
Denmark-based Novo Nordisk’s Wegovy tablets and U.S.-based Eli Lilly’s Foundayo have led the shift from injectable weight loss medications to oral ones.
In response, Korean pharmaceutical companies have increased their efforts to commercialize their weight loss medications in global markets, with a focus on how easy it is to take the medication, as convenience has become a key factor in determining a product’s success.
Foundayo, made with orforglipron, has been on sale in the United States since Monday, five days after the U.S. Food and Drug Administration gave its approval. The medication falls under the category glucagon-like peptide-1 receptor agonists (GLP-1), a hormone that plays a key role in regulating blood sugar levels and can suppress a person’s appetite.
The Wegovy tablet is made of semaglutide, which, like Foundayo, can suppress a person’s feelings of hunger by stimulating insulin production and inhibiting glucagon. The medication was launched in the U.S. market in January.
Both Foundayo and Wegovy come in the form of pills, and one is meant to be taken daily.
However, while the Wegovy tablet must be taken on an empty stomach in the morning, Foundayo can be taken before or after eating — a fact that Eli Lilly highlights in its advertising.
Foundayo showed a weight reduction rate of 12.4 percent in the 72nd week, and the Wegovy pill demonstrated a 16.6 percent reduction at the 64th week, according to clinical trial data from the respective manufacturers.
![Novo Nordisk's Wegovy is on display at a product launch event in a hotel in Seoul on Oct. 15, 2024. [YONHAP]](https://koreaseafood.online/data/photo/2026/04/13/f8670e0b-16b8-4f17-86dd-709e8f8ad6b5.jpg)
Novo Nordisk's Wegovy is on display at a product launch event in a hotel in Seoul on Oct. 15, 2024. [YONHAP]
Both are sold at the same price in the U.S. market.
With private insurance coverage, U.S. patients can access the lowest-dose versions of both medications for $25 per month. The uninsured price starts at $149 per month.
Eli Lilly has submitted applications to sell Foundayo in more than 40 countries and plans to launch the product worldwide upon approval. The medication is expected to take one to two years to reach the Korean market.
“As edible obesity treatments do not require a cold chain supply route — as their injectable counterparts must be transported at low temperatures — they are more convenient, which translates into their relatively affordable prices,” said an economic research center at Koreabio, a biotechnology association.
“This year is likely to become the ‘year of oral obesity treatments,’” the center added.
Meanwhile, Korean pharmaceutical companies are accelerating the speed with which they develop treatments, with a focus on differentiation.
![A notice stating that Wegovy is in stock is posted on the window of a pharmacy in central Seoul on Dec. 2. [YONHAP]](https://koreaseafood.online/data/photo/2026/04/13/4c871295-531c-4610-9b0d-4baf4cf08d51.jpg)
A notice stating that Wegovy is in stock is posted on the window of a pharmacy in central Seoul on Dec. 2. [YONHAP]
For example, Hanmi Pharmaceutical plans to launch its GLP-1 injectable efpeglenatide — reportedly tailored to Koreans’ obesity rates — in the second half of this year; Ildong Pharmaceutical is preparing to enter the second phase of clinical trials for an oral obesity treatment; and Daewoong Pharmaceutical is developing a patch-type obesity drug that painlessly delivers the medication through the skin.
They are also preparing next-generation obesity treatments.
Celltrion is developing a “quad-action injectable” and a “multiaction oral drug” at the same time, while Dx&Vx is researching an improved small-molecule GLP-1-based oral obesity treatment. Quad action refers to medication that works on four different targets simultaneously.
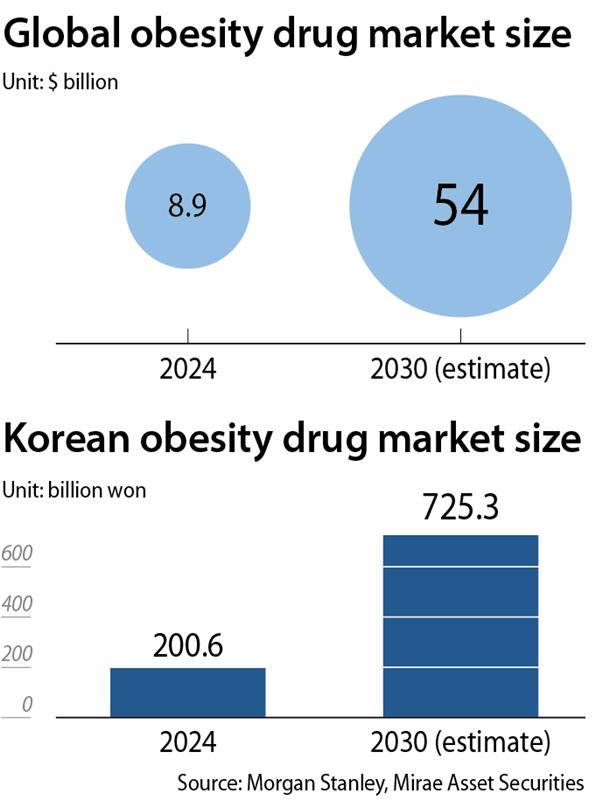
Korea’s pharmaceutical industry is optimistic about the global outlook of domestic obesity drugs, given that the worldwide overweight and obese population exceeds 1.7 billion.
According to the medical market surveyor IQVIA, the global obesity treatment market reached $46.07 billion in 2025, an 82 percent increase from the previous year. Korea is the fifth-largest obesity treatment market in the world, following the United States, Brazil, Canada and Australia.
“Obesity care is likely to become part of routine health management due to the launch of weight loss pills and medication,” Koreabio said. “The market is expected to expand further through differentiated formulations.”
This article was originally written in Korean and translated by a bilingual reporter with the help of generative AI tools. It was then edited by a native English-speaking editor. All AI-assisted translations are reviewed and refined by our newsroom.
BY KIM KYUNG-MI [[email protected]]










with the Korea JoongAng Daily
To write comments, please log in to one of the accounts.
Standards Board Policy (0/250자)